How many moles are in PCl3?
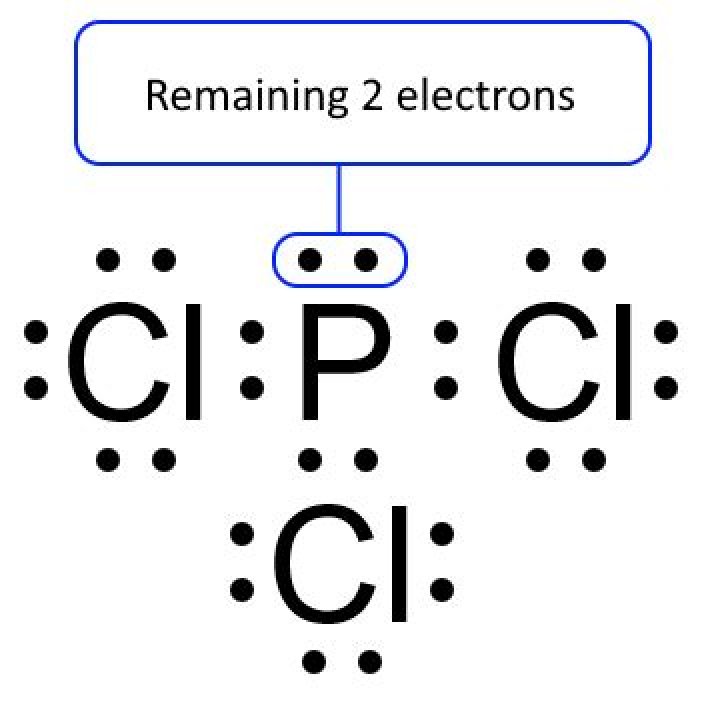
We assume you are converting between moles PCl3 and gram. You can view more details on each measurement unit: molecular weight of PCl3 or grams This compound is also known as Phosphorus Trichloride. The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles PCl3, or 137.332761 grams.Click to see full answer. Moreover, how do you find the molar mass of PCl3? Find the molar mass of PCl3 Answer. elizabethabuh20. 137.33 g/mol. Formula is PCI3. Density is 1.57 g/cm2. answers left. Join for free. Click to let others know, how helpful is it. Furthermore, how many molecules are in a mole? Avogadro’s number is a very important relationship to remember: 1 mole = 6.022×1023 6.022 × 10 23 atoms, molecules, protons, etc. To convert from moles to atoms, multiply the molar amount by Avogadro’s number. To convert from atoms to moles, divide the atom amount by Avogadro’s number (or multiply by its reciprocal). Consequently, how do you find the number of moles? Use the molecular formula to find the molar mass; to obtain the number of moles, divide the mass of compound by the molar mass of the compound expressed in grams.What is the mass ratio of pcl3? Percent composition by element Element Symbol Mass Percent Chlorine Cl 77.446% Phosphorus P 22.554%


